Financial Report & Highlights 2021

As treasurer of our society, I am proud that we have endured the financial risks faced during the global COVID-19 pandemic and its extensive worldwide impact. After the coronavirus breakout in 2020, a contingency plan was developed to secure staff positions and strategic investments. This has allowed us to invest in our strategic mission in 2021 while building some financial reserves in order to face present and future uncertainties. We were able to close 2021 with a positive financial result and EBMT continues to invest in its strategic goals in line with its mission.
The EBMT organisational structure and creation of income for scientific and educational projects is pictured below. Moreover, in 2021 the EBMT obtained again an “unqualified opinion” from an independent auditor, affirming that EBMT is maintaining its high standard of modern management, sustaining its financial stability, and spending allocating money to our mission.
Spending financial resources on our missions
Our steady annual income has been used to strengthen our growing organisation as a whole. For 2021, the budget needed for management and administration (i.e., Board and Executive Office expenses) stabilised to 19.8% which allocates 80.2% to our scientific studies, registry, accreditation and education like the 2021 Annual Meeting, E-learning and educational events. Therefore, EBMT remains a very efficient organisation when assessed by international rankings (“A” according to CharityWatch).
Continuing with balanced financial roadmap
In 2021, we continued to secure and improve our staff positions while further optimising EBMT organizations through a well-balanced financial roadmap for the future of EBMT. Our goal was to be able to cover all financial challenges in line with our ambitions but also provide a strong backbone for the organisation. The EBMT Board decided to partially release allocated reserves in 2021 (409,000€) to support our organisation for the next few years which covers the following topics and areas.
-
Continuing with virtual meetings
Due to the ongoing COVID-19 pandemic we organised for the second time our annual congress as a virtual meeting while vaccination campaigns were ongoing. Thanks to the great work and dedication of the Barcelona office, the 2021 virtual annual meeting was a great success because we were able to maintain the high level of education, the support of our sponsors and the engagement of our delegates. The completed in-housing operations of successive EBMT educational events strengthens the EBMT’s educational positioning and visibility within the virtual arena and by having financial control.
-
Adapting the organisational structure
In 2021, we further adapted the managerial infrastructure of the EBMT which is structured in six departments Registry, Clinical Research, Education & Events, Advocacy & Quality of Care, Sales & Development and Support. Each department is led by a department Director who, together with the Executive Director and Medical Officer, form the EBMT Management Team.
In 2021 we continued with improving the Clinical Research, Sales and Support departments in order to increase and streamline the process of setting up membership, sponsorship, retrospective and prospective studies in collaboration with pharma, institutions and non-profit organisations as well as marketing & communications. The sponsorships and funded studies are a growing segment of the activities of EBMT and important financial pillar for our strategic mission. Due to the solid financial situation of EBMT, we were able to sustain all staff positions during the ongoing COVID-19 pandemic and finance the new, key positions within the transformed EBMT organisational structure. Further streamlining and restructuring of the organigram will continue in 2022 in order to increase the efficiency of EBMT.
-
COVID Taskforce and science fund
A COVID task force within EBMT was formed to support patients and the EBMT community during the COVID-19 pandemic. All working parties were involved in the COVID task force which was very productive in developing general and vaccination guidelines for patients, donors and the EBMT community, scientific surveys and Covid-19 related scientific studies. The EBMT Board decided to strategically allocate reserves in a science fund to support the Covid-19 studies and for strategic scientific studies in next years. In education and events contract management a Covid-19 clause was incorporated to minimize the inherited financial risk.
-
Continuing the CAR T PASS Studies
After receiving the qualification opinion from EMA regarding the cellular therapy module of the EBMT Registry, the Board decided to raise manpower to support this initiative, which will collect post-market evidence for safety and efficacy of different commercial CAR T products. Contracts with three big pharma companies have been signed and more companies are likely to follow. The finance support behind these contracts allows for more human resources to execute these PASS studies and increase the Registry data and data quality. This registry tool will be helpful for all researchers executing clinical studies with immune effector cells to gather more comprehensive data sets.
-
Updating the EBMT Registry
In 2021, we kept working on building the future Registry into a new IT system while continuing to enter transplant and cellular therapy data in ProMISe. Furthermore, we built a new CASTOR database to enter CAR T data for the EBMT and PASS scientific studies. Building of the future Registry platform has the highest financial priority. Reserves have been earmarked to cover all expected costs. In addition, reserves are being used and held for any unexpected expenses. Furthermore, the London, Leiden and Paris offices were and are still closely working together on this project under the supervision of the Registry Director, and the new Deputy Executive Director for the Registry upgrade.
-
JACIE Remote Inspections
In 2021, JACIE deployed a system for remote inspections and as an optional format for re- accreditations including IEC inspection. Remote inspections have made it possible to carry out inspections that would had to be cancelled due to the risk of contagion. In addition, JACIE has also implemented a hybrid inspection model with part of the inspection team onsite and others by remote inspection. Following the forced stop to activity due to the Covid-19 pandemic, JACIE was pleased to resume on-site inspections during the last quarter of 2021. Further streamlining and restructuring formats will continue in 2022 in order to increase the efficiency of JACIE accreditation.
Financial conclusion
The ongoing positive financial development of the organisation over the past years has allowed EBMT to further build on its strategic goals and improve its organisational structure. Current highlights include the successful virtual meetings during the COVID-19 pandemic, increasing E-learning opportunities, registry upgrade, cellular therapy registry, benchmarking system, CAR T PASS studies, staff compensation and human resource management, information technology innovation, COVID-19 studies and the Science fund. In spite of the financial challenges related to the ongoing COVID-19 pandemic, EBMT will be closing the year 2021 with a positive total result of 2,008 k€ (expenses of 8,013 k€ and a total income of 10,021 k€) thanks to the fantastic work of the EBMT staff, community and the use of our financial reserves.
It has been my pleasure to serve and guide EBMT as Treasurer in 2021 during the global COVID-19 pandemic, and I am excited to continue my tasks in 2022 while prospects for the world to open up again are positive. I would like to express my appreciation for the finance team in the Leiden office, Bas Natkiel, Liesbeth Hoekstra and Erwin Timmers for their hard work and dedication.
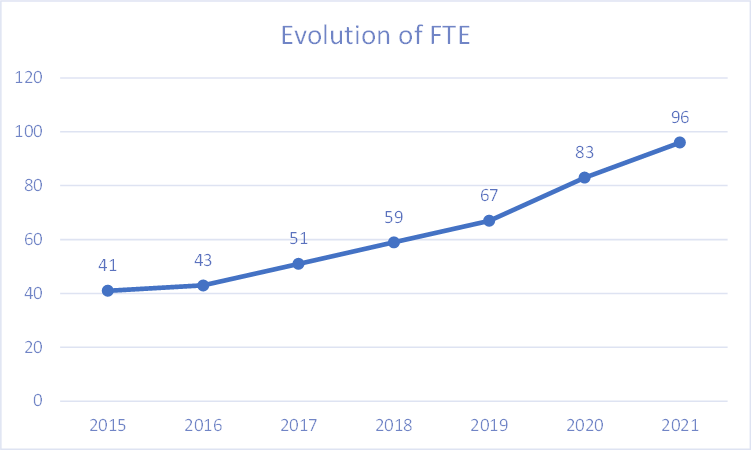
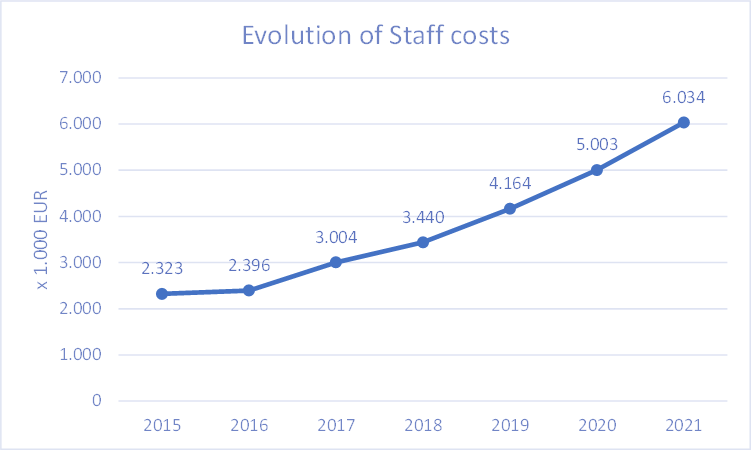
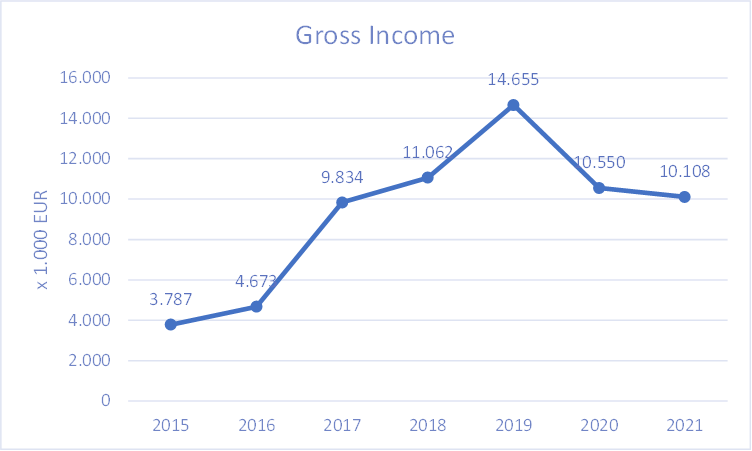
Financial Highlights 2021
SOURCE OF INCOME |
EUR | % |
DESTINATION OF RESOURCES |
EUR | % | |
|---|---|---|---|---|---|---|
| EBMT MEMBERS DONATIONS | 603,000€ | 6.0% | SCIENTIFIC STUDIES | 3,400,000€ | 42.4% | |
| SCIENTIFIC SPONSORS DONATIONS | 575,000€ | 5.7% | EBMT REGISTRY | 610,000€ | 7.6% | |
| ANNUAL MEETING | 3,962,000€ | 39.5% | ANNUAL MEETING | 579,000€ | 7.2% | |
| RESULTS FROM INVESTMENTS | 1,000€ | 0.1% | EDUCATIONAL ACTIVITIES | 1,283,000€ | 16% | |
| STANDARDS & ACCREDITATION (JACIE) | 558,000€ | 7.0% | ||||
| NON-EARMARKED INCOME | 5,141,000€ | 51.3% | TOTAL MISSION COST | 6,430,000€ | 80.2% | |
| GRANTS FOR STUDIES, CLINICAL TRIALS & EDUCATION | 4,623,000€ | 46.1% | MANAGEMENT AND ADMINISTRATION | 1,583,000€ | 19.8% | |
| ACCREDITATION (JACIE) | 257,000€ | 2.6% | ||||
| EARMARKED INCOME | 4,880,000€ | 48.7% | ||||
| TOTAL INCOME | 10,021,000€ | 100.0% | TOTAL EXPENDITURE | 8,013,000€ | 100,0% |
FINANCIAL OUTCOME |
EUR | % |
|---|---|---|
| TOTAL INCOME | 10,021,000€ | 100.0% |
| TOTAL COSTS | 8,013,000€ | 80,0% |
| GROSS RESULTS | 2,008,000€ | 20.0% |
| EARMARKED | 409,000€ | 4,1% |
| NET RESULT | 1,599,000€ | 15.9% |
At the time of the report release date (March 17, 2022), the audit process is pending completion
