
Title: Severe CNI-Induced Hypertriglyceridemia with Neurological Hyperviscosity After HSCT
Submitted by Angeliki Georgopoulou, Dimitrios Tsokanas and Maria Liga, Department of Hematology, BMT and Cellular Therapy Program, University of Patras, Patras, Greece
Physicians expert perspective: Alexandros Spyridonidis
A 47-year-old woman with FLT3-ITD–positive acute myeloid leukemia (AML) relapsed 3.6 years after a first sibling donor allogeneic hematopoietic stem cell transplantation (allo-HSCT). The first transplant course was uncomplicated; she received sorafenib maintenance for two years and remained in good general health until relapse. She proceeded to a second allo-HSCT from a different donor in complete remission. Conditioning included thiotepa, melphalan, and treosulfan. GVHD prophylaxis consisted of alemtuzumab and cyclosporine.
On day +67, she developed stage III acute cutaneous GVHD that resolved with prednisone. Concurrently, she reported persistent headache and was found to have severe hypertriglyceridemia (1,603 mg/dL, normal <150 mg/dl), attributed to cyclosporine. A similar episode of hypertriglyceridemia during the first transplant in late 2021/2022 resolved after cyclosporine discontinuation, and the subsequent baseline lipid profile in the in the subsequent years remained within acceptable limits and did not require lipid-lowering therapy. (Figure 1)
Cyclosporine was therefore replaced with tacrolimus. One week later, she developed mild acute disorientation without hemodynamic instability. MRI and cerebrospinal fluid analysis excluded infectious and structural etiologies; fundoscopy showed no papilledema or hemorrhage. Triglycerides rose to 4,357 mg/dL, and hyperviscosity syndrome secondary to extreme hypertriglyceridemia was diagnosed.
Urgent plasmapheresis and insulin infusion led to rapid neurological recovery and a rapid decline in triglyceride levels. Tacrolimus was discontinued and replaced with mycophenolate mofetil; ruxolitinib was later added for recurrent stage II skin GVHD. Five months post–second transplant, she remains in complete remission, without GVHD, with a normal lipid profile and excellent clinical condition.
Figure 1. Serum triglyceride levels from the time of the first transplant (November 2021) onward.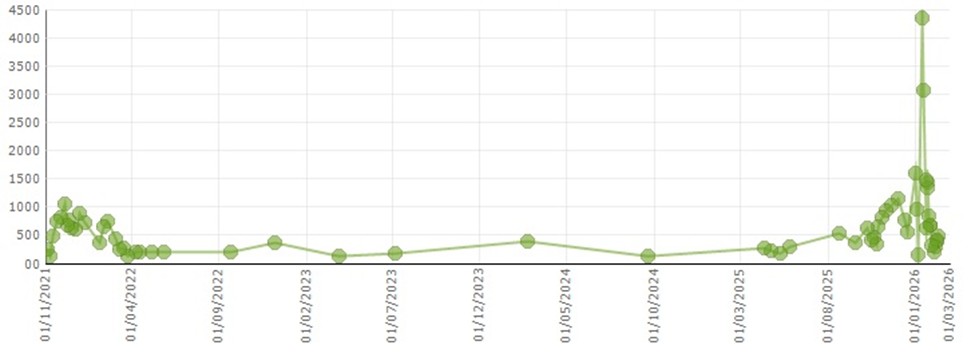
Q. In an allo-HSCT recipient presenting with acute neurological symptoms and severe hypertriglyceridemia (e.g., >4,000 mg/dL), after exclusion of infection and structural CNS causes, what is the most appropriate immediate intervention?
A. High-dose statin therapy and observation
B. Fibrate therapy only, with dietary fat restriction
C. Insulin infusion ± urgent plasmapheresis
D. Continue the calcineurin inhibitor
Expert Perspective
This case highlights a rare but severe manifestation of calcineurin inhibitor (CNI)–associated metabolic toxicity in the allo-HSCT setting. The patient developed recurrent, extreme hypertriglyceridemia, first with cyclosporine and subsequently after switching to tacrolimus, culminating in symptomatic hyperviscosity syndrome. The course underscores individual susceptibility, the possibility of cross-toxicity between CNIs, and the potentially life-threatening consequences of post-transplant dyslipidemia.
Dyslipidemia is common after allo-HSCT, particularly within the first 3–6 months, reflecting a convergence of systemic inflammation, reduced activity, nutritional changes, and immunosuppressive therapy. CNIs and corticosteroids are major pharmacologic contributors. Cyclosporine is a recognized cause of hypertriglyceridemia and mixed dyslipidemia, often characterized by increased triglycerides (predominantly VLDL), with concomitant elevations in LDL and total cholesterol.
Cyclosporine affects lipid homeostasis through multiple mechanisms involving both hepatic handling and peripheral clearance. It inhibits LPL, reducing triglyceride hydrolysis in chylomicrons and VLDL, which leads to hypertriglyceridemia. It also decreases bile acid synthesis and interferes with LDL receptor function, contributing to elevated cholesterol levels. Additionally, cyclosporine induces insulin resistance and β-cell dysfunction; because insulin stimulates LPL and suppresses hepatic VLDL production, insulin resistance further worsens hypertriglyceridemia. Concomitant corticosteroids can amplify these effects by increasing hepatic VLDL production and reducing peripheral lipid clearance.
In this patient, recurrence of severe hypertriglyceridemia across two transplants strongly suggests an underlying predisposition, despite a post–first transplant lipid profile that did not warrant lipid-lowering therapy and no prominent conventional metabolic risk factors. The reproducible temporal relationship and resolution upon cyclosporine withdrawal support causality.
The subsequent dramatic rise in triglycerides to 4,357 mg/dL after tacrolimus initiation raises important clinical considerations. Although tacrolimus is generally associated with less severe dyslipidemia than cyclosporine, owing to weaker effects on lipoprotein lipase activity and cholesterol transport, clinically significant lipid abnormalities may still occur, particularly when combined with corticosteroids. In patients with prior calcineurin inhibitor (CNI)–associated dyslipidemia, switching between CNIs may not mitigate metabolic risk. The rapid and extreme increase observed in this case suggests that calcineurin inhibition itself, rather than a cyclosporine-specific mechanism, was sufficient to destabilize lipid homeostasis in a susceptible host.
Clinically, triglyceride levels above 1,000–2,000 mg/dL increase pancreatitis risk; however, this case illustrates a less frequent but critical complication: hyperviscosity syndrome presenting with neurological manifestations. Extreme hypertriglyceridemia increases plasma viscosity through chylomicron accumulation, impairing cerebral microcirculation and leading to neurological symptoms such as headache, confusion, and disorientation. Notably, fundoscopy may be normal; absence of retinal findings does not exclude clinically relevant cerebral hyperviscosity.
The decision to initiate urgent plasmapheresis was appropriate. Plasmapheresis rapidly removes triglyceride-rich lipoproteins, reduces viscosity, and restores microvascular flow, consistent with the prompt neurological recovery after two sessions. Intravenous insulin infusion is an effective adjunct by enhancing LPL-mediated clearance, even in normoglycemic patients when glucose is co-administered.
This case highlights key management principles: (1) early recognition and frequent lipid monitoring in high-risk patients, particularly during the first months post-HSCT; (2) prompt modification of the causative agent when clinically feasible; and (3) escalation to urgent apheresis and metabolic therapy when severe symptoms or extreme triglyceride levels occur. For longer-term GVHD control in patients with severe CNI metabolic toxicity, non-CNI–based strategies may be required, as illustrated by the eventual use of mycophenolate mofetil (a drug that has not been shown to have any direct effects on lipoprotein metabolism or induce hyperlipidemia) and ruxolitinib in this patient.
Finally, beyond acute risk, dyslipidemia contributes to late cardiovascular morbidity in transplant survivors. Structured cardiometabolic surveillance integrated into survivorship care is essential, especially in patients who experience severe early metabolic events.
Correct Answer:
C. Insulin infusion ± urgent plasmapheresis
Rationale: Extreme hypertriglyceridemia can cause symptomatic hyperviscosity with neurological manifestations. When severe symptoms occur and triglycerides are markedly elevated, plasmapheresis provides the fastest reduction of triglyceride-rich lipoproteins and plasma viscosity. Insulin infusion further accelerates triglyceride clearance by enhancing lipoprotein lipase (LPL) activity. Dietary measures and oral lipid-lowering drugs are important for longer-term control but are too slow for acute hyperviscosity.
Key Take-Home Messages
- Cyclosporine, and to a lesser extent tacrolimus, can induce severe hypertriglyceridemia via impaired lipoprotein clearance and insulin resistance. Mycophenolate mofetil is less dyslipidemic.
- Risk is highest early post-transplant and with concomitant corticosteroids.
- Extreme hypertriglyceridemia may present with neurological manifestatons due to hyperviscosity.
- Acute management requires rapid triglyceride reduction (insulin ± plasmapheresis) and modification of immunosuppression.
- Long-term cardiometabolic surveillance is important in HSCT survivors.
References:
- Bis G, Szlasa W, Sondaj K, Zendran I, Mielcarek-Siedziuk M, Barg E. Lipid complications after hematopoietic stem cell transplantation (HSCT) in pediatric patients. Nutrients. 2020;12(9):2500.
- Miller LW. Cardiovascular toxicities of immunosuppressive agents. Am J Transplant. 2002;2(9):807–818.
- DeFilipp Z, Duarte RF, Snowden JA, et al. Metabolic syndrome and cardiovascular disease following hematopoietic cell transplantation: screening and preventive practice recommendations from CIBMTR and EBMT. Bone Marrow Transplant. 2016;52:173–182.
- de Groen PC. Cyclosporine, low-density lipoprotein, and cholesterol. Mayo Clin Proc. 1988;63(10):1012–1021.
- Griffith ML, Savani BN, Boord JB. Dyslipidemia after allogeneic hematopoietic stem cell transplantation: evaluation and management. Blood. 2010;116(8):1197–1204.
- Subramanian S, Trence DL. Immunosuppressive agents: effects on glucose and lipid metabolism. Endocrinol Metab Clin North Am. 2007;36(4):891–905.
- Lu Y, Ma X, Pan J, Ma R, Jiang Y. Management of dyslipidemia after allogeneic hematopoietic stem cell transplantation. Lipids Health Dis. 2022;21(1):65.
Future Clinical Case of the Month
If you have a suggestion for future clinical case to feature, please contact Anna Sureda.