JACIE Activity report 2025


2025 marks a step change for JACIE as it continued with a high level of operational activity whilst transitioning into a new governance model. None of this work would be possible without the active and committed community of Centres and the Inspectors contributing their time and expertise to the benefit of the JACIE community.
Governance change: from a Committee to a Working Party
The EBMT General Assembly voted for JACIE to become a Working Party (WP) in the Annual Meeting 2025 and hence, for the first time, JACIE now has a permanent representation at the EBMT’s governing body – Scientific Council and the Board of Association. Following the change to a Working Party status, JACIE has amended its governance structure to fit within the WP framework. The Working Party Board includes representatives from all JACIE Committees and Task Forces. In addition, JACIE WP was delighted to welcome Dr Joaquim Vives as the ISCT representatives to the WP Board. He takes up his role after Dr Fermin Sanchez-Guijo Martin stepped down in summer 2025.
JACIE WP also said goodbye to Dr Lynn Manson as the first WP chair in October 2025 with Dr Charles Crawley stepping up as an interim WP Chair.
Restructure of the governance structure whilst maintaining oversight on JACIE’s operational activities has been challenging, but has also opened up opportunities to discussion a wider publication and educational strategy for JACIE.
Activity update
JACIE continued its strong performance in inspection and certification. This was only possible due to the commitment and sheer hard work by the inspectors and the members of the Assurance (Accreditation) Committee.
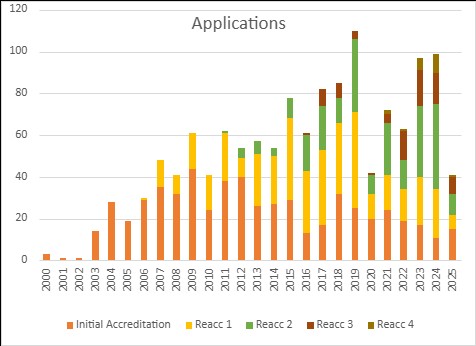
The number of applications received by JACIE reduced significantly in 2025. This is due to the impact of Covid in 2021-22. As the certification cycle is four years, centres due to apply for re-certification was understandably low in 2025.
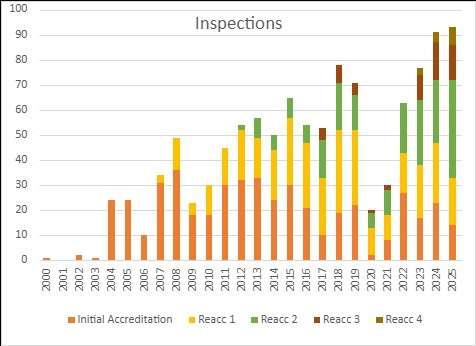
Inspection activity remained strong and JACIE carried out 93 inspections in 2025 (91 in 2024) matching the number of inspections in 2024. In addition, JACIE collaborated in three joint inspections with FACT under Stepwise initiative in Latin America. JACIE also expanded its geographical reach with first JACIE inspections in India and China.
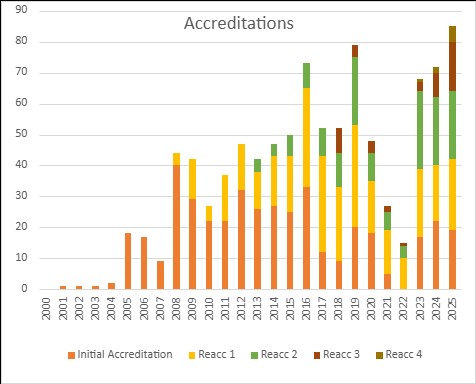
The high level of inspection activity for the last two years was also reflected in the increase in the number of certifications in 2025. Total of 84 centres completed the certification process in 2025 in comparison to 71 in 2024. JACIE WP Board thanks all the members of the Assurance (Accreditation) Committee meeting fortnightly to review and provide oversight to the inspection and reporting processes and enhancing objectivity of the overall process.
Publication of the 9th Edition of HCT and 3rd Edition of IEC Standards
FACT and JACIE published the 9th Edition of the FACT–JACIE International Standards for Hematopoietic Cell Transplantation (HCT) and the 3rd Edition of the International Standards for Immune Effector Cells (IEC) in October 2025. These updated Standards represent a major milestone in the continuous effort to promote quality, safety, and excellence in cellular therapy worldwide, reflecting the rapid scientific, clinical, and regulatory developments in the field.
EBMT and JACIE extend sincere gratitude to the Standards Development Sub-Committee co-chairs and the committee members for their outstanding commitment, expertise, and collaborative spirit throughout this extensive revision process. Their rigorous review, thoughtful debate, and dedication ensured that the Standards remain scientifically robust, clinically relevant, and globally applicable, while continuing to support centres in delivering the highest levels of patient care.
The publication of these new editions would not have been possible without the substantial voluntary contributions of time and knowledge from all involved including the centres and the individuals who responded to the public consultation. EBMT and JACIE warmly thank everyone who contributed to this important achievement and acknowledges their essential role in advancing quality standards for hematopoietic cell transplantation and immune effector cell therapies worldwide.
Inspector activities
In 2025, three Inspector Training Courses were delivered in Rome, Italy; Bern, Switzerland, and Berlin, Germany, in conjunction with Centro Nazionale Trapianti (CNT), Swiss Blood Stem Cell Transplantation and Cellular Therapy (SBST), and Deutsche Arbeitsgemeinschaft für Hämatopoetische Stammzelltransplantation und Zelluläre Therapie (DAG-HSZT) respectively. Through these collaborations, a total of 75 new inspectors were trained. Inspector training days remain a highlight of the JACIE calendar, and we are grateful to our partner organisations for their continued support and commitment to strengthening the inspector community.
JACIE also continued its grants programme to support inspectors’ participation in the 51st Annual Meeting of the EBMT, held in Florence, Italy. The initiative continues to provide valuable opportunities for learning, networking, and collaboration, and remains a much valued Inspector incentive. In total, 77 grants were awarded, enabling inspectors to attend the meeting either in person or virtually.
Institutions accredited in 2025
| Facility | Institution | City | Country |
|---|---|---|---|
| Programma Trapianto di CSE | POLICLINICO RIUNITI – AZIENDA OSPEDALIERO UNIVERSITARIA | Foggia | Italy |
| Pediatric Stem Cell Transplantation Programme | Tel Aviv Sourasky Medical Center | Tel Aviv | Israel |
| Centro Trapianti Ferrara | Azienda Ospedaliero Universitaria di Ferrara | Cona (Ferrara) | Italy |
| Hématologie – Thérapie cellulaire – Besançon CHU et EFS | Centre Hospitalier Universitaire (CHU) Besançon Etablissement Français du Sang Bourgogne Franche-Comté (EFS BFC) | Besançon | France |
| Cambridge and Ipswich BMT and Cellular Therapy Programme | Addenbrookes Hospital | Cambridge | United Kingdom |
| Skåne University Hospital, Lund | Skåne University Hospital Lund | Lund | Sweden |
| Haematopoietic Stem Cell Transplantation & CAR-T Cell Therapy Program (Adult) | University Hospital Würzburg, Department of Internal Medicine II, Centre for Haematopoietic Stem Cell Transplantation & CAR-T Cell Therapy | Würzburg | Germany |
| BMT Programme and IEC Therapies | Nottingham University Hospital NHS Trust | Nottingham | United Kingdom |
| Programa de Trasplante Hematopoyético y Terapias CAR-T | Hospital Universitario Donostia | San Sebastian | Spain |
| Servicio de Hematología del Hospital Universitario Marqués de Valdecilla – Unidad de Terapias Avanzadas del Banco de Sangre y Tejidos de Cantabria | Hospital Universitario Marqués de Valdecilla - Fundación Marqués de Valdecilla | Santander | Spain |
| Birmingham Centre for Cellular Therapy and Transplant (BCCTT) | Birmingham Centre for Cellular Therapy and Transplant (BCCTT), University Hospitals Birmingham NHS Foundation Trust | Birmingham | United Kingdom |
| MTN - Milano Transplant Network | Fondazione IRCCS Cà Granda Opsedale Maggiore Policlinico - University of Milano Presidio ospedaliero Fatebenefratelli, ASST Fatebenefratelli-Sacco | Milan | Italy |
| Unità Operativa Complessa di Ematologia e Terapia Cellulare | Istituto Tumori “Giovanni Paolo II” – IRCCS | Bari | Italy |
| HPC Transplantation Programme, University Hospital Centre Zagreb | University Hospital Centre Zagreb, Clinical Department of Transfusion Medicine and Transplantation Biology | Zagreb | Croatia |
| Gesundheit Nord gGmbH, Klinikum Bremen-Mitte, Zentrum für Innere Medizin, Medizinische Klinik 1 | Gesundheit Nord gGmbH, Zentrum für Innere Medizin, Medizinische Klinik 1 | Bremen | Germany |
| Department of Pediatric Oncology and Hematology, Charité – Universitätsmedizin Berlin | Charité Universitätsmedizin Berlin | Berlin | Germany |
| Thérapie Cellulaire et Hématologie Clinique Adulte | Service de Thérapie Cellulaire et d’Hématologie Clinique Adulte - CHU ESTAING | Clermont-Ferrand | France |
| Alberts Cellular Therapy (ACT) | Alberts Bouwer and Jordaan Inc. | Pretoria | South Africa |
| Hospital Universitario Gregorio Marañón | Hospital Universitario Gregorio Marañón | Madrid | Spain |
| STEM CELLS PROGRAMME - OSPEDALE SAN RAFFAELE | I.R.C.C.S. OSPEDALE SAN RAFFAELE | Milan | Italy |
| HEMATOPOIETIC STEM CELL TRANSPLANT AND CELLULAR THERAPY. HOSPITAL RAMON Y CAJAL | HOSPITAL UNIVERSITARIO RAMON Y CAJAL | Madrid | Spain |
| Programma Trapianti Congiunto Adulto Pediatrico | Clinica di Ematologia, Azienda Ospedaliero Universitaria delle Marche | Ancona | Italy |
| TPH-Pediátrico Hospital de la Santa Creu I Sant Pau (HSCSP) | Hospital de la Santa Creu i Sant Pau | Barcelona | Spain |
| Programme de greffe de cellules souches hématopoïétiques et de thérapie cellulaire | Hôpitaux Universitaires de Genève | Geneva | Switzerland |
| Cellular and Molecular Therapies (CMT) | NHS Blood and Transplant | Birmingham | United Kingdom |
| Centro Trapianti Alessandria | AOU Alessandria | Alessandria | Italy |
| Adult Stem Cell Transplantation & Cellular Therapy Programme | Azienda Ospedaliero-Universitaria Careggi (AOUC) | Florence | Italy |
| University Hospitals Coventry and Warwickshire | University Hospitals Coventry and Warwickshire - NHS Trust | Coventry | United Kingdom |
| Dept.of Haematology and Oncology | Charles University Hospital Pilsen | Pilsen | Czech Republic |
| Institute of Haematology and Blood Transfusion | Institute of Haematology and Blood Transfusion | Prague | Czech Republic |
| Sheffield Blood & Marrow Transplant and Cellular Therapy Programme | Sheffield Blood & Marrow Transplant and Cellular Therapy Programme - Royal Hallamshire Hospital | Sheffield | United Kingdom |
| Programma Trapianti Cellule Staminali | G.O.M. “Bianchi-Melacrino-Morelli" | Reggio Calabria | Italy |
| Cellular Therapy Programme | Universitair Ziekenhuis Brussel (UZ Brussel) | Brussels | Belgium |
| Programma Trapianti di Cellule Staminali Ematopoietiche, Terapia Cellulare e Terapia Genica | Ospedale Pediatrico Bambino Gesù | Rome | Italy |
| Klinischer Transplantationsbereich, Universitätskliniken und Polikliniken für Innere Medizin IV und Pädiatrie I | Universitätsklinikum Halle (Saale), Einrichtung für Transfusionsmedizin | Halle | Germany |
| Birmingham Women’s and Children’s NHS Foundation Trust | Birmingham Women's and Children's NHS Foundation Trust | Birmingham | United Kingdom |
| Hematopoietic Transplant Programe, Hematology Department | Hospital Universitario La Princesa | Madrid | Spain |
| Wits Donald Gordon Cellular and Immunotherapy Centre (WDG CITC) | Wits Donald Gordon Medical Centre | Johannesburg | South Africa |
| Oncohematology Department | University Hospital “ San Giovanni di Dio e Ruggi D’Aragona” Salerno | Salerno | Italy |
| Hématologie Immunologie et Oncologie pédiatrique, CHU TIMONE ENFANTS | AP-HM CHU TIMONE ENFANTS | Marseille | France |
| Transplantation Unit, Department of Pediatric Hematology and Oncology. University Hospital Motol, Prague | Motol and Homolka University Hospital | Prague | Czech Republic |
| Welsh Bone Marrow Donor Registry | Welsh Blood Service (WBS) | Pontyclun | United Kingdom |
| Copenhagen Bone Marrow Transplantation Program | Copenhagen University Hospital Rigshospitalet | Copenhagen | Denmark |
| Children’s Health Ireland (CHI) | at Crumlin | Dublin | Ireland |
| Trapianti Cellule Staminali Emopoietiche | Ospedale Sant’Eugenio - ASL Roma 2 | Rome | Italy |
| Klinischer Transplantationsbereich, Universitätskliniken und Polikliniken für Innere Medizin IV und Pädiatrie I | Universitätsklinikum Halle (Saale) | Halle | Germany |
| Programma Trapianto CSE- AOUS | UOC Terapie Cellulari e Officina Trasfusionale- AOUS | Siena | Italy |
| London Bridge Hospital, Private Care at Guy’s | London Bridge Hospital | London | United Kingdom |
| Hammersmith Hospital | Imperial College Healthcare NHS Trust | London | United Kingdom |
| Unidad de Hematología y Hemoterapia | Hospital General Universitario Morales Meseguer – Centro Regional de Hemodonación de Murcia | Murcia | Spain |
| Stem Cell Transplantation Program, Haga Hospital, Department of Hematology | HagaZiekenhuis | The Hague | Netherlands |
| Cell Processing Centre, Department for Therapeutic Services | Slovenian Institute for Transfusion Medicine | Ljubljana | Slovenia |
| Cellular and Molecular Therapies (CMT) | NHS Blood and Transplant | Bristol | United Kingdom |
| Programma Trapianto Autologo e Terapie Cellulari – Ospedale Nuovo | ASST Ovest Milanese – Ospedale Legnano | Legnano | Italy |
| Service Hematologie Clinique et Therapie Cellulaire | Hôpital Saint-Antoine – Assistance Publique – Hôpitaux de Paris | Paris | France |
| Klinikum Stuttgart | Klinikum Stuttgart | Stuttgart | Germany |
| Stem Cell Transplantation Programme Maastricht UMC+ | Maastricht University Medical Center | Maastricht | Netherlands |
| Erciyes University Pediatric BMT Center | Erciyes University Medical Faculty Pediatric BMT Center | Kayseri | Turkey |
| PROGRAMA DE TRASPLANTE Y TERAPIA CELULAR DEL COMPLEXO HOSPITALARIO UNIVERSITARIO DE VIGO | COMPLEXO HOSPITALARIO UNIVERSITARIO DE VIGO | Vigo | Spain |
| Policlinico Tor Vergata – SIMT PTV | Metropolitan Rome Transplant Program: RTN – Team 1 Policlinico Tor Vergata | Rome | Italy |
| BMT UNIT Frankfurt (Oder), Klinikum Frankfurt (Oder) GmbH | Klinikum Frankfurt (Oder) GmbH, Medizinische Klinik I | Frankfurt (Oder) | Germany |
| Ematologia e Trapianto CSE Azienda Ospedaliera “Card. Panico” Tricase (LE) | Azienda Ospedaliera “Card. Panico” Tricase (LE) | Tricase | Italy |
| University Children´s Hospital, Dept. of Paediatric Haematology/Oncology, Paediatric Stem Cell Transplantation Program | University Medical Center Würzburg | Würzburg | Germany |
BMT Unit Oncology Center “Marianna V. Vardinogianni – Elpida’’ Aghia Sophia Children’s Hospital | SCT and Cellular Therapies Unit, Pediatric Oncology Center “Marianna V. Vardinogianni – Elpida’’ | Athens | Greece |
| HTCT - Hematology Transplant and Celullar Theraphy | UZ - Leuven Campus Gasthuisberg Hematology | Leuven | Belgium |
| Struttura Complessa di Ematologia con Trapianto di CSE e DIMT | Laboratorio di Ematologia /Presidio Ospedaliero S.G. Moscati - P.O.C. ASL/TARANTO | Taranto | Italy |
| Pediatric Hemto-Oncology, BMT and Cell Therapy Ward | The Edmond and Lily Safra Children's Hospital_ Sheba Medical Center | Ramat-Gan | Israel |
| University Medical Center Hamburg-Eppendorf | Department of Stem Cell Transplantation, Division of Pediatric Stem Cell Transplantation and Institute of Transfusion Medicine | Hamburg | Germany |
| The Royal Marsden NHS Foundation Trust Transplant Programme | The Royal Marsden Hospital NHS Foundation Trust | London (Sutton) | United Kingdom |
| Programa de Trasplante Pediátrico de Progenitores Hematopoyéticos | Hospital Universitario Vall d’Hebron | Barcelona | Spain |
| Klinik und Poliklinik für Innere Medizin C | University of Greifswald | Greifswald | Germany |
| HEMATOPOIETIC STEM CELL TRANSPLANTATION ISTITUTO G. GASLINI | IRCCS ISTITUTO G. GASLINI | Genova | Italy |
| Programma Trapianti Udine | Clinica Ematologica ed Unità Terapie Cellulari - Azienda Sanitaria Universitaria Friuli Centrale (ASUFC) | Udine | Italy |
| GUSTAVE ROUSSY CELL THERAPY PROGRAMME (Programme de Thérapie Cellulaire de Gustave Roussy Cancer Campus) | Gustave Roussy | Villejuif | France |
| Sheffield Children’s NHS Foundation Trust HSCT Programme Haematology and Oncology Department | Sheffield Children’s Hospital | Sheffield | United Kingdom |
| BMT Center Erciyes University Medical School, Dept. of Hematology | Erciyes University Kapadokya (Cappadocia) | Kayseri | Turkey |
| The Christie NHS Foundation Trust | The Christie NHS Foundation Trust | Manchester | United Kingdom |
| Universitätsklinikum Freiburg, Klinik für Innere Medizin I, Hämatologie, Onkologie und Stammzelltransplantationen | Universitätsklinikum Freiburg, Klinik für Innere Medizin I, Hämatologie, Onkologie und Stammzelltransplantationen | Freiburg | Germany |
| Adult Haematology, Stem Cell Transplant & Cellular Therapy Department - Riyadh | King Faisal Specialist Hospital & Research Centre | Riyadh | Saudi Arabia |
| Programma Trapianti di CSE Lecce | UO Ematologia PO Vito Fazzi | Lecce | Italy |
| The Hematology Facility from the St Antonius Ziekenhuis | St. Antonius Hospital Nieuwegein, St Antonius Ziekenhuis and Sanquin Blood Supply | Nieuwegein | Czech Republic |
| Alder Hey Children’s NHS Foundation Trust | Alder Hey Children’s, Ward 3B, NHS Foundation Trust | Liverpool | United Kingdom |
| Programma Trapianti Metropolitano Adulti di Cellule Staminali Emopoietiche Bolzano-Trento | Azienda sanitaria dell’Alto Adige/Südtiroler Sanitätsbetrieb, Comprensorio Sanitario di Bolzano & Azienda Provinciale per i Servizi Sanitari Trento Struttura Dipartimentale di Ematologia Ospedale S. Chiara TN | Bolzano | Italy |
| Apheresis Collection – Division of Hematology, Luzerner Kantonsspital, Luzern | Luzerner Kantonsspital | Luzern | Switzerland |
Committee activities
Throughout the year, the Inspector Committee (JIC) organised and delivered five Inspectors’ webinars, offering important updates on the Standards and support to inspectors on the inspection process. These webinars were available to all inspectors live, and continue to be available as recorded sessions, ensuring broad access and ongoing engagement across the community.
JACIE Quality Managers Committee (QMC) co-published a paper with the GOCART Coalition in November 2025. The paper is based on a survey conducted in January 2025 among hospitals across Europe and beyond, yielding responses from 146 institutions. View the publication "Operational burden and fragmented implementation in CAR T-cell therapy: Insights from a multinational survey by the GoCART Coalition and the JACIE Quality Managers Committee"
In addition, the QMC also debuted their first podcast on 18 December 2025, continuing the series into 2026.
