The EBMT Registry


In 2025, 37.329 new patients were registered in the EBMT Registry. 18.875 new allogeneic HCTs were reported, 18.504 autologous HCTs, and 4.757 CAR-Ts. These trends are consistent with previous years.
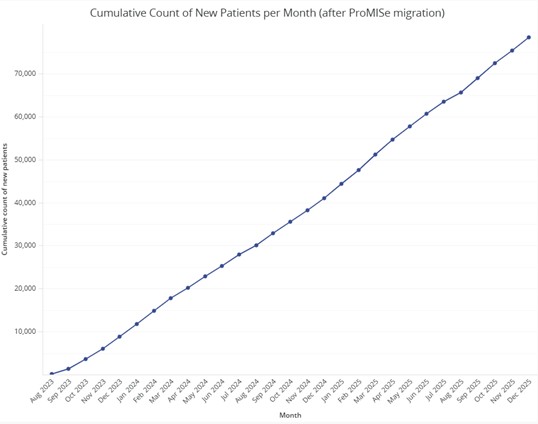
In total, the EBMT Registry had 804.745 patients on December 31, 2025. Patients can be classified into 13 diagnosis classifications.
| Diagnosis classification | Count of diagnosis events |
|---|---|
| Acute leukaemia | 218.155 |
| Autoimmune disease | 5.155 |
| Bone Marrow Failure incl. Aplastic Aneamia | 25.550 |
| Chronic leukaemia | 34.296 |
| Hemoglobinopathy | 10.562 |
| Inborn Errors | 13.520 |
| Lymphoma | 220.622 |
| Myelodysplastic syndrome | 30.438 |
| Myelodysplastic/myeloproliferative disease | 7.422 |
| Myeloproliferative disorder | 12.904 |
| Other Diagnosis | 2.669 |
| Plasma cell neoplasm | 212.450 |
| Solid tumour | 51.864 |
| Total diagnosis events | 845.607 |
EBMT Registry
In October 2025, EBMT achieved ISO/IEC 27001:2022 certification, demonstrating the EBMT Registry adheres to the international standards for safely storing and processing data.
The EBMT Registry itself had seven releases that introduced new functionality. With the addition of summary fields on patients, an audit trail, and export functionality, it has been made easier for users to quickly access any data they need. Several technical updates were made for improved system and study management. MicroStrategy, the exporting tool launched in 2024, has been migrated to an optimised cloud environment with improved performance and additional functionality. On top of that, ten new reports were added to the library.
A new EBMT megafile has been developed to enable effective reporting from the registry, focusing on working-party-initiated studies.
2025 also marked the year the EuroTraCTOR grant closed, which started in 2022 and has been a cornerstone of the EBMT Registry's development through collaboration with the consortium partners.
Studies
In the first half of the year, the EBMT Registry Study Manager was being piloted with two studies from Working Parties. After a successful evaluation, additional functionalities were added, and more studies are being rolled out. The Study Manager tool will allow Working Parties to manage all study-related information, including additional data collection, inside one platform to improve efficiency on studies.
Communication
Every eight weeks, representatives from the Registry Unit met with the national registries to inform them about ongoing developments and projects. In these meetings the national registry stakeholders also provided the Registry Unit with valuable feedback on the EBMT Registry and projects. The Registry Committee held monthly meetings to discuss ongoing issues and difficulties reported from various stakeholders. Next to this, all EBMT Registry users received monthly newsletters with updates.
Team
The Registry Unit acquired one new IT analyst in 2025, who works from the Barcelona office. The Registry Unit now consists of one manager, one IT team lead, one data operations team lead, one data management specialist, four IT analysts, three data managers, and one data steward. The team is located in Spain and the Netherlands.
Future
In 2026, the Registry Unit's goals are centred around stability and consolidation of good practices, implementing strong data governance policies together with data quality procedures to ensure the data provided by EBMT’s members can be used to support EBMT’s mission of saving and improving the lives of patients with blood-related disorders. Updated data collection forms are being developed with a planned launch in April 2026, after the Annual Meeting. The aim is to focus on the important variables of the core dataset and moving several variables to the extended dataset. This will allow focussing on EBMT studies and reducing the workload of the centres reporting data.
